English Resume: Give Evidence about Examples for Chemical Reaction in Everyday Life
Examples for Chemical Reaction in Everyday
Life
Chemical reactions can happen
anywhere around us, not just in the laboratory. Matter interacts to form new
products through a process called chemical reactions or chemical changes. Every
time we cook or are cleaning, it is also a chemical in action. Our bodies live
and grow thanks to chemical reactions. There are reactions when we take drugs,
light a match, and take a breath. Here are 10 examples of chemical reactions in
everyday life. This is just a small example, because we see and experience
hundreds of thousands or even more chemical reactions every day.
1.
Burning
Every
time we light a match, burn a candle, make a fire, or light a grill, we will
see a burning reaction. Combustion combines energetic molecules with oxygen to
produce carbon dioxide and water.
For
example, propane combustion reactions, found in gas grills and some fireplaces,
are:
C3H8 + 5O2 → 4H2O
+ 3CO2 + Energy
2.
Corrotion
Rust is
iron oxide, usually a red oxide formed by redox reactions of iron and oxygen in
the presence of water or air humidity. Some forms of rust are distinguished
both visually and by spectroscopy, and form under different circumstances. Rust
consists of hydrated iron (III) oxide Fe2O3 · nH2O
and iron (III) oxides-hydroxides (FeO (OH), Fe (OH)3).
In
sufficient time, oxygen, and water, every iron mass will eventually convert
entirely of rust and crumbling. The rust surface is flaked and brittle, and
does not provide protection to the base iron, such as the formation of patina
on the surface of the copper. Rust is a general term for corrosion of iron and
its alloys, such as steel. Many other metals have equal corrosion, but the
resulting oxide is not commonly called rust.
Here is
the chemical equation for iron rust:
Fe + O2 + H2O → Fe2O3.
XH2O
3.
Mixing of
Chemicals
If we
just combine vinegar and baking soda to make chemical volcanoes or milk with
baking powder in a recipe, we experience the displacement or double reaction
metathesis (plus some other things). The material recombines to produce carbon
dioxide and water gases. The form of carbon dioxide bubbles in the volcano and
can help increase roasting.
These
reactions seem simple in practice, but often consist of several steps. Here is
the overall chemical equation for the reaction between baking soda and vinegar:
HC2H3O2 (aq)
+ NaHCO3 (aq) →
NaC2H3O2 (aq) + H2O (l) + CO2
(g)
4.
Batteries
Batteries
are electrical-chemical devices that store energy and emit energy in the form
of electricity. A battery usually consists of three important components,
namely:
Ø The
carbon rod as anode (the positive pole of the battery)
Ø Zinc (Zn)
as cathode (negative pole of battery)
Ø Pasta as
electrolyte (conductor)
Batteries
use electrochemical or redox reactions to convert chemical energy into
electrical energy. Spontaneous redox reactions occur in galvanic cells, while
chemical reactions do not occur spontaneously in electrolytic cells.
5.
Acid and
Bases Reaction
Whenever
we mix acid (eg, vinegar, lemon juice, sulfuric acid) with a base (eg, baking
soda, soap, ammonia, acetone), we do acid-base reactions. This reaction
neutralizes acids and bases to produce salt and water.
Sodium
chloride is not the only salt that can be formed. For example, here is the
chemical equation for an acid-base reaction that produces potassium chloride, a
substitute for common table salt:
HCl + KOH → KCl + H2O
6.
Soaps and
Detergens
Soaps and
detergents can be cleaned by using chemical reactions. Soap emulsifies dirt,
which means soap binds stain oil stains so they can be cleaned with water. The
detergent acts as a surfactant, lowering the surface tension of the water so it
can interact with oil, isolate them, and and clean it.
References
https://bisakimia.com
https://en.wikipedia.org

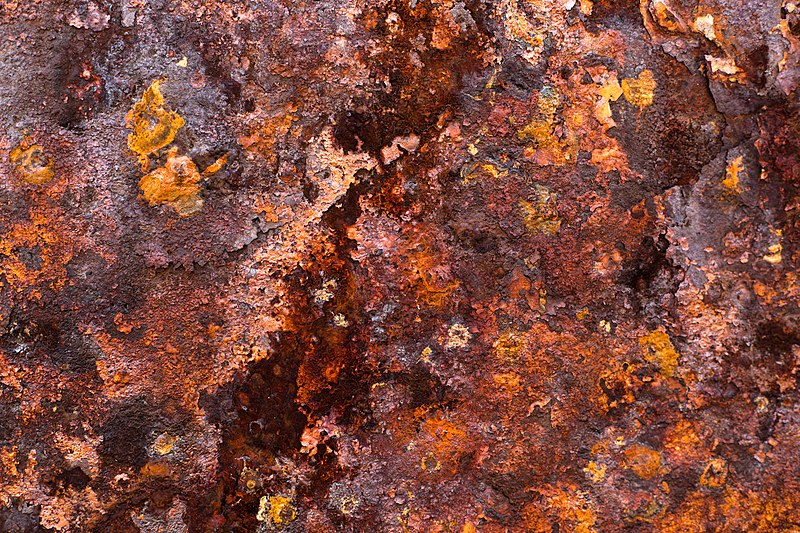





Describe to me about chemical reaction dor soap ? Please
BalasHapusSoap is one of the oldest known chemical compounds. The soap itself is never actually found, but comes from the development of a mixture of alkaline and fat / oil compounds.
HapusA soap made by carboxilic acid, alcohol, and ester.
http://amrita.olabs.edu.in/userfiles/1/image/img8.png
hi linggo , can you give me more example of this topic ?
BalasHapuslike evidence of chemistry reaction in our body , or system like respiration etc , thanks
Photosynthesis: 6 CO2 + 6 H2O + Light → C6H12O6 + O2
HapusAerobic Cellular Respiration: C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy (36 ATPs)
Anaerobic Cellular Respiration: C6H12O6 → 2C2H5OH + 2CO2 + Energy
Give examples of adverse chemical reactions in daily life
BalasHapusFor example of adverse chemical reaction in daily life is use lead or Pb in car or motorcycle engine. With high Lead (Pb) content, in the long run it will affect the level of human emotions that breathe through breathing, And can lead to low level of IQ in children. Use premium gasoline is Tetra Ethyl Lead (TEL). With tis reaction;
HapusTEL is produced by reacting chloroethane with a sodium–lead alloy.[9]
4 NaPb + 4 CH3CH2Cl → (CH3CH2)4Pb + 4 NaCl + 3 Pb
TEL is also produced by reacting Ethylmagnesium Iodide with lead(II) chloride.
4C2H5Mg-I + 2PbCl2 → (C2H5)4Pb + Pb +4 MgICl
The product is recovered by steam distillation, leaving a sludge of lead and sodium chloride. TEL is a viscous colorless liquid. Because TEL is charge neutral and contains an exterior of alkyl groups, it is highly lipophilic and soluble in petrol (gasoline).
Is there any way to distinguish acid and base without using indicator? Explain
BalasHapuslinggo,Why aluminum metal is resistant to corrosion compared to iron ?
BalasHapuswhat it is corrosion and what th effect of corrosion?
BalasHapusHow the chemical reaction can happen?
BalasHapusDoes all iron have corrosion? And how long it will cause corrosion?
BalasHapus